C. javanus Kieffer 1924Synonyms: All suggested synonyms are doubtful as most probably refer to C. vitellinus. Chironomus prasinellus - Tokunaga 1940 (misidentified) but Sasa & Kikuchi, 1995. Chironomus vitellinus Freeman 1961 (Chaudhuri et al. 1992) - incorrect synonymy (see Chironomus vitellinus). Yamamoto (2002) has suggested that this species should be in a separate subgenus Austrochironomus, as type of the subgenus. However there is doubt that there is a consistent set of characters for the species he included. There is no meaningful nearest neighbor to this bin. Most specimens in the Bin are actually C. vitellinus, with only a few C. javanus. C. javanus and C. vitellinus are closely related although there are clear morphological differences between them (Martin & Epler 2024). Adult: Female. Yellow. Eyes separated by not more than their terminal width, gradually thinning at the top. Palps long, brownish black, 4th segment matching the previous two segments combined, 2nd shorter than 3rd, 1st much longer than wide [these are actually segments 2-5]. Antenna 2nd segment narrowed in the middle, the neck a little longer than wide, the rest broken. Metanotum, three short bands, mesonotum and mesonotum reddish. Halteres light green. Wing whitish, not distinctly stippled, veins a whitish yellow, crossvein and base of the cubital black, cubital arched, ending very near the tip of the wing. Legs light green, fore tarsus long and thin, white, both ends of segments 1-4 deep black, 5th slightly clouded, pulvilli a little wider, with long hairs, not exceeding the middle of the crotchets, hardly shorter than the empodium, probably branched four hind tarsi broken; fore femur much longer than the tibia, the latter and the tarsal segments are 2 : 3 2/3 ; 2 : 1 1/2 : 2 : 3/4 [i.e. LR = 1.80], the 4th segment is longer than 3rd, the four hind tibias have confluent combs which occupy two thirds of the circumference, the two spurs short. Abdomen a bright green, unmarked. L. 4 mm. Male. Pale yellow, abdomen spotless, four bands on mesonotum, metanotum and mesosternum fawn, red scape, flagellum broken. Wing as female. Legs white, distal end of tarsomeres 1-4 and 5th tarsal segment black. Anterior tarsus broken. Eyes separated by 1.5 times their terminal width. Terminal articles of the genitalia ('pince') arcuate, the distal half suddenly narrowed in a straight beak, glabrous, having only one third of the width of the proximal half and carrying on the distal half of the medial side straight six large rigid bristles. Superior appendages very thin, glabrous, linear, reaching the end of the basal article (gonocoxite), weakly curved and ending in a point; inferior appendages large, pubescent, exceeding just the gonocoxite and bearing dorsally the usual long and thick curved setae. Anal point long and thin. L. 4.5 mm. Many specimens attributed to C. javanus are actually C. vitellinus and C. javanus appears to have a relatively restricted distribution in India, Java, and Malaysia. A yellowish-green species with dark bands on the tarsi and darkening of the cross veins of the wings. Other than the original description, the only reliable further descriptions for this species are those of Johannsen (1932) and Chaudhuri et al. (1992) (whose specimens appear to be larger, possibly due to being reared in laboratory), plus a specimen from Penang and another from India. Measurements can vary considerably, which may be due to different means of measuring or due to variations in the climate in which the specimen has developed. Wing: length 1.96 (1.92-2.03) mm, width 0.62 (0.56-0.63) mm; VR 0.94 (0.92-0.95); brachiolum with 1 or 2 SCf; 14 (8-18) setae in squamal fringe. Head: Frontal tubercles present, abt 32x18 µm (1.8 times longer than wide); about 16.8 (14-22) clypeal setae; AR 3.33 (2.70-3.82). Palpal proportions (segs. 1-5): 42 : 36 : 155 : 166 : 260; P5/P4 1.38-1.58; P5/P3 1.53-1.60. Thoracic setae: abt 8-10 acrostichals; 14.3 (12-16) dorsocentrals; 4.3 (2-6) prealars; 1 supraalar; abt 5 (3-7) small setae in anterior row, about 9 larger setae in posterior row; total 10 (8-12). Leg proportions and ratios (micron):
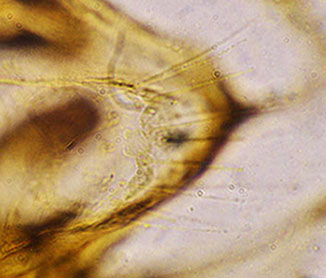 TIX showinng the rows of setae Note the abdomen is squashed towards the bottom right, and the two brown masses at left are the displaced IVo's. Hypopygium with long tubular anal point, strongly turned down. Superior volsella well developed and curved, perhaps closest to E(h) of Strenzke (1959); Inferior volsella stout, reaching just beyond the end of the Superior volsella, about 1/3 of gonostylus length, with 12-14 simple incurved setae. Gonostylus quite swollen and conspicuously narrowed over posterior third to half, with about 1+2+1 or 1+4+1 setae at tip. 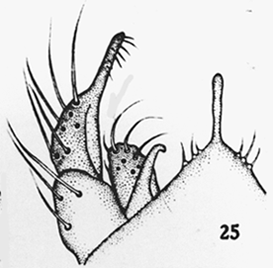 Illustration of the hypopygium of C. javanus From Johannsen (1932) Female  Pupa and Fourth instar larva: See C. javanus immatures. Cytology: The chromosomes of C. javanus have not been studied, but there is a photograph which is probably this species (below): Four polytene chromosomes with the thummi-cytocomplex combination AB, CD, EF, G. Centromeres are obvious but not heterochromatic. Essentially terminal nucleolus in arm G, possibly with a BR close to it. A large puff is developed about one third from the distal end of arm B. No reported polymorphism.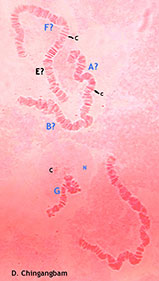 Molecular: The mitochondrial CO1 barcode sequence is available in GenBank and the BOLD database for a few specimens. Although the sequences in BOLD are in a single Bin, there is a suggestion that the distribution is bimodal, with the few C. javanus sequences separated to the right of the main group. The BARCODE sequences differ by about 47 base positions (this does not include some rare polymorphisms where one or two C. vitellinus specimens carry the sequence present in C. javanus), and the majority are in the 5' region (as is common for differences between closely related Chironomus species):  The present measurements differ in some significant details from those of Kieffer's description, although the other details of colour and the unusual genitalia correspond. The males are easily recognised by the unusual genitalia, particularly the tubular anal point, and the larvae by the 6-toothed premandible. The pupa is a fairly typical for Chironomus.Males can be separated from the relatively similar C. vitellinus by the tubular anal point rather than expanded at the distal end (see below ) and the placement of the setae on TIX, although that species has been regularly misidentified as C. javanus as can be readily seen from the figures of the male hypopygium. The larvae can be separated by the number of teeth on the premandible (6 vs. 7 – so the excellent photograph of the premandible in Cranston (2007) is C. vitellinus, not C. javanus), the unusually short 3rd segment of the antenna (shared with C. vitellinus) and the teeth of the mandible (type II-IIIA vs type IIIC). Found:Type locality - Buitenzorg, Java, INDONESIA, also Sumatra (Johannsen 1932). Apparently limited distribution through India, Java, Malaysia, and parts of Africa, in rice paddies and small temporary water bodies, even sewage works. The redescription of adults and immatures from Malaysia by Al-Shami et al. (2012) clearly refers to C. vitellinus but they did also collect at least one specimen of C. javanus (figure above). |
Modified: 5 July 2025
Access: Unrestricted
Copyright © 2005-2025, Jon Martin.