Species 2l. C. pallidivittatus sensu Beermann (1955) |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
| | | | | | | | | | | |
Apparently no setae at center of TIX.
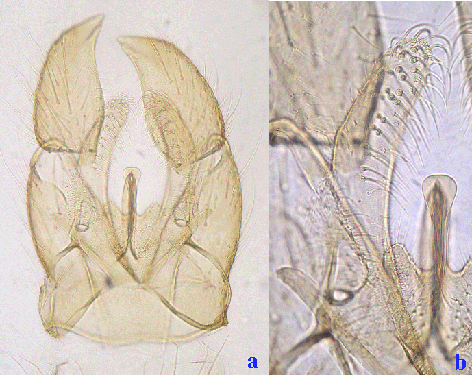
a. whole hypopygium; b. superior appendage, shorter than inferior appendage.
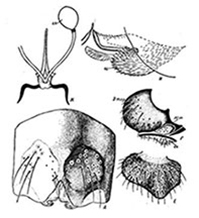
Female: Some characters of European specimens have been described in Russian by Shilova (1978). The illustrations of the genital region are below.
Pupa: Length (male) about 16.5 mm; inner margin of wing case about 1.64 mm. Cephalic tubercles about76x78 µm, seta at least 50 µm.; lacking 2ndary cephalic tubercles and frontal warts.
Basal ring about 177x89 µm, with core waisted in the middle; HR 1.99.
Hook row of abdominal segment II with about 65 recurved hooks. Hook row about 0.4 mm wide, segment width 1.14 mm, so hook row occupies about 35% of segment width. Large Pedes spurii B on segment III, small one on segment II; large Pedes spurii A on segment IV, small ones on segment V and VI.
Postero-lateral spur of eighth segment relatively fine, with one or two spines.
About 81 tainiae on each side of the swim fin.
Larva a medium to large plumosus-type (fem. 18.0-19.7 mm; male 14.3-17.2 mm); lateral tubules developed 422 (340-680) µm); Anterior ventral tubules (1.64 (1.24-2.60) shorter than posterior pair (1.81 (1.28-3.08); those of males generally shorter..
Gular region generally pale but occasionally darkened, frontoclypeus darkened, at least slightly, posteriorly.
Mentum (fig. c) with relatively pointed teeth; 4th laterals about in line with other teeth; c1 tooth moderately broad with parallel sides, c2 teeth well developed and sharp pointed (type IIA).
Ventromentum about 250 µm wide and 65 µm deep (figs d and f), with deep clefts on inner edge of the marginal region; abt 52-55 VMR abt 0.32-0.35.
Premandible (fig. b) with outer tooth about 5 times the width of the inner (tooth Ty.B2), teeth coming to fine points. Pecten epipharyngis (fig. a) with about 13-14 even sharp teeth.
Antenna (fig. g) with RO about a third up from the base, A1 about 4 times longer than wide and abt 4.7 time longer than A2, AR abt 2.15, A3 about same length as A4 and longer than A5; segment proportions (µm): 167 : 35 : 13 : 13 : 8.
Distance between antennal bases about the same as that between the S4 setae.
Mandible (fig. e) with third inner tooth generally dark and well separated (type IIB-IIIC), about 24-25 furrows on outer surface at base; Mdt-Mat about 38 µm; MTR 0.38.
Cytology: 4 pairs of chromosomes with the camptochironomus arm combination AB, DE, CF, G. Arm G closely paired with three Balbiani rings, 2 close together near one end with a constriction between most distal and the end, and the other towards the other end. Nucleolus in arm B, virtually at centromere, but other smaller ones may be developed distal to this in sequence B2, known only as a heterozygote and apparently confined to males (i.e. male determiner on arm B).
pal h'A1: 1ab, 7d-7b, 3i, 2c-1h, 9c-8f, 10-9d, 11-12, 3h-2d, 8e-a, 1c-g, 7a-4, 13-19
pal n'B8:
pal n'B9: (sex-linked)
pal h'C2: 1a-e, 15-14d, 4-6b, 8g-a, 2e-3c, 11c-9, 1f-2d, 11d-14c, 19-16, 7-6c, 20-22
pal h'D2: 1a-2b, 15-14, 22-18e, 8-9, 17-18d, 16a-e, 11-13, 3g-2c,7-4, 10e-a, 23-24
pal n'D3:
pal h'E1: 1-2g, 4b-f, 12f-10c, 3f, 8g-10b, 3a-e, 8f-4c, 12g-13
pal h'F1: 1a-d, 9b-12, 3b-2f, 13-14c, 5d-6, 9a-7a, 14d-16, 5c-3c, 1e-2e, 17-23 i.e. as h'F1 of tentans
pal n'G4:
pal n'G5:
Found: ¿Ontario - South March (45.24°N, -75.52°W), Carleton Co.
Manitoba - Lake Winnipeg (Sæther 2012).
Saskatchewan - 5ml nw Theodore.
North Dakota - Cleveland
South Dakota - Vermillion (42.47°N, -96.46°W), Clay Co.; Sioux Falls, Minnehaha Co.; Lake Francis Case, Gregory Co.
(43.27°N, -99.00°W).
Generally at low frequency in prairie sloughs containing C. dilutus
Hein and Schmulbach (1971) first described this species in North America, noting that it was not the same as C. pallidivittatus Johannsen. The cytology of North American populations in comparison to that of European populations was described by Kiknadze et al. (1998a).
Kiknadze et al. (1991) noted that larvae of the European populations of C. pallidivittatus could be distinguished from the related C. tentans by the smaller size as reflected in the head width and the length of antennal segment 1. It is not clear whether this could be applied to separating the North American specimens from those of C. dilutus, particularly in view of the occurrence of hybridization and the existence of different races of the latter species.
C. pallidivittatus and C. dilutus cannot be separated on the basis of the DNA "barcode" sequence of cox1, but can be separated by the sequence of the globin gene gb2β (Martin et al. 2002).
It appears that the male determiner is on arm B, as in C. tentans and C. dilutus.
Modified: 2 March 2025
Access: Unrestricted
Copyright © 2000-2025, Jon Martin.