NEW ZEALAND and PACIFIC CHIRONOMUS SPECIES
Larval types and Karyotypes, with Adults where data available
Integration of data of Jon Martin1 and Don Forsyth2
1 jonmartin8@bigpond.com
2 Deceased
| When Freeman (1959) revised the New Zealand Chironomidae he recognized two species of the genus Chironomus (s.s.): C. zealandicus and C. analis. However, even at this stage it was known that adults of C. zealandicus could be produced from two distinct larval types, a salinarius-type (lacking ventral tubules) or a so-called "thummi-type" (with ventral tubules)(Forsyth, 1971), although in fact the latter are a bathophilus-type. (The current classification system can be found here.) Analysis of the polytene chromosomes further indicated that there were different chromosome number forms of both the salinarius- and the thummi-type larvae ( Lentzios et al. 1980), which appeared to be distinct species. With the integration of the morphological and ecological analyses of Don Forsyth, the karyotypic studies of Jon Martin, and DNA studies with Ian Hogg and Sofia Ibarrarán, we have concluded that there are at least fourteen species of Chironomus in New Zealand. (Note that this only includes one of the two species described by Sublette & Wirth (1980) from the subantarctic islands), and includes an Australian species that may be a recent migrant. DNA studies are suggesting some of these are groups of closely related species as is common in other parts of the world. Where possible a link to the relevant Bin in the BOLD Database (http://www.boldsystems.org/index.php) is given.
Seven of the currently recognised species have a salinarius-type larva, and seven others are bathophilus-type, although one is polymorphic for ventral tubule development, some having only slight development of the posterior pair of ventral tubules (halophilus-type). The larva of the fifteenth species is unknown. For convenience, some Pacific island species are also included (see below). |
This listing is also available in pdf format (7.6 MB)(Updated 23 February 2025).
In general, the morphological terminology used in this document follows Sæther (1980), Webb & Scholl (1985) and Vallenduuk & Moller Pillot (1997).
Provisional Key to Adult Males
(based on data of D.J. Forsyth) | 1a | Anal point stout | C. analis | | 1b | Anal point narrower | 2a | | | | | | 2a(1b) | Abdominal tergites with dark saddle spots | 5a | | 2b | Abdominal tergites with bands or patches covering most of the segment | 3a | | | | | | 3a(2b) | Anterior tergites with a band over only about two thirds of the segment | 4a | | 3b | Anterior tergites with band over most of the segment | C. 'thermarum' | | | | | | 4a(3a) | Pigmentation of the anterior tergites narrowing to the posterior edge | C. 'castaneum' (North Island) | | 4b | Pigmentation of anterior segments covering whole width of segment | 7a | | | | | | 5a(2a) | Anterior LR generally above 1.5; only short sparse beard | C. forsythi (part) | | 5b | Anterior LR below 1.5; may or may not have beard | 6a | | | | | | 6a(5b) | Lacking a beard | C. 'paracastaneum' | | 6b | Dense short beard and sparse long beard | C. zealandicus | | | | | | 7a(4b) | Anterior LR generally above 1.5; only short sparse beard (BR <3) | C. forsythi (part) | | 7b | Anterior LR probably just below 1.5, at least some longer beard (BR 3-7) | 8a | | | note: *C. nr. antipodensis would also fit here, but LR not known | | | | | | 8a(7b) | Dense short beard and sparse longer beard (BR 4-7) TIX setae in single patch | C. novaezelandiae | | 8b | Moderate beard (BR 3-4) TIX setae in individual patches | C. sp. NZ12 | Note that the adults of some species are unknown.
The adults of C. 'spilleri', are assumed to be similar to those of C. novaezelandiae since Don Forsyth included them all under that name.
The Australian species C. 'pseudoppositus' has been recognized in New Zealand on the basis of mtCOI sequence, but the morphology has not been compared with that of Australian specimens. If similar, LR same as C. forsythi. There is also the large reared female, C. sp.NZ3a. SV typeIn the adult descriptions reference is made to the types of superior volsella shape as recognized by Strenzke (1959). This is a helpful initial classification, but experience has shown that the types are not discrete, but are part of a continuum. The three categories as described by Strenzke are:
S-type: The SV is shoe shaped, i.e. it is drawn out distal-medially into a broad, rounded lobe (Fig. a-c, below) (Strenzke's figure suggests the most distal point will be at the toe of the shoe).
D-type: The SV is ribbon-like: distally it may have a weakly thickened shoulder (Fig. d, below) (most distal point is not at the internal margin), or bent in a shallow sickle-shape (Fig. e-f, below).
E-type: The SV has the form of an elephant's tusk; distally it is sharply graded to a point, or with an expanded knob (Fig. g-i, below) (line from base to most distal point goes outside the limits of the SV).

Provisional Key to Fourth Instar Larvae | 1a | Larva a salinarius-type | 2a | | 1b | Larva with at least some development of ventral tubules | 8a | | | | | | 2a(1a) | More than 60 striations on Ventromental plate | C. zealandicus (= C. species a) | | 2b | Less than 60 striations on Ventromental plate | 3a | | | | | | 3a(2b) | Anal tubules relatively long sometimes with slight constriction near middle | C. forsythi in part | | 3b | Anal tubules relatively short, pointed or rounded | 4a | | | | | | 4a(3b) | Frontoclypeal region of head pale or only slightly darkened | 5a | | 4b | Frontoclypeal region very dark | 6a | | | | | | 5a(2a) | c1 tooth of mentum broad (IIA) and flattened | sp. NZ9 | | 5b | c1 tooth of mentum narrower (type IIA) and more pointed | C. 'thermarum' | | | | | | 6a(4b) | c1 tooth of mentum broad (type IIA) | 7a | | 6b | c1 tooth of mentum generally narrower.(usually type 3) | C. analis | | | | | | 7a(6a) | Mandible of ty. IIB or C (occasionally IIIC), with about 15-25 furrows | C. 'castaneum' and C. 'campano' | | 7b | Mandible of ty. III, with about 26 furrowsC | sp. NZ14 | | | | | | 8a(1b) | Slight development of posterior ventral tubules only | C. 'thermarum' (in part) | | 8b | Two pairs of ventral tubules present | 9a | | | note: for 8a, in thermal waters (but C. zealandicus, species 10 and sp. 12 can be also) | | | | | | | 9a(8b) | Anal tubules relatively long, as in C. forsythi | 10a | | 9b | Anal tubules shorter and rounded | 11(a) | | | | | | 10a(9a) | Anal tubules all about equal and 2.5x longer than wide (3 polytene chromosomes) | sp. NZ8C. sp. NZ8 | | 10b | Anal tubules about 2.5-4.2 times longer than wide, dorsal pair often longer. | C. 'pseudoppositus' | | | | | | 11a(9b) | Head somewhat narrower, ratio mentum width/VHL less than 0.65, ventral tubules sometimes longer than 1.0 mm. | C. novaezelandiae group | | | Note: Frontoclypeus generally darkened but sometimes pale or only slightly darkened (which group would include Ty. 2; C. "spilleri"; and C. sp. NZ12). Variability possibly reflecting presence of more than one species. | | | 11b | Head broader, ratio mentum/VHL above 0.65; with darkened frontoclypeus, ventral tubules about 0.5-1.0 mm. | C. sp. NZ7 | | | Note: not in North Island | | There is also the larva of the reared female, C. sp.NZ3a. Reference is also made to the mentum and mandible types originally devised by Webb & Scholl (1985) and Vallenduuk & Moller Pillot (1997) and Proulx et al. (2013), as well as a ventromental plate character. The previous classifications were made for relatively small numbers of species, but they do not cover all the variability seen in these characters and so further modification has been necessary. The mentum type is defined only by the degree of development of the 4th lateral teeth:

Type I - height in same line as the rest of the lateral teeth;
Type II - 4th laterals reduced, height about equal to that of the 5th laterals;
Type III - 4th laterals further reduced, height less than that of the 5th laterals.The mentum may be further classified by the characters of the central trifid tooth:
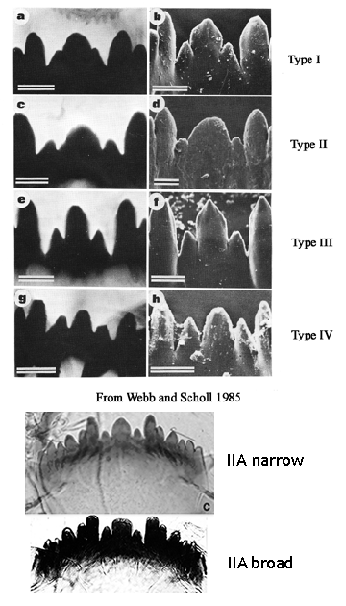
Type IA - c2 teeth only partially separate from c1, i.e. as shoulders on c1 (figure a).
Type IB - c2 teeth slightly more separated (figure b).
Type IIA - c1 broad, c2 teeth distinctly separated (figure c).
Type IIB - c1 very broad, c2 less separated (figure d).
Type III - c1 tooth relatively narrow and much higher than the separated c2 teeth (figs e and f).
Type IV - c2 teeth well separated, not much lower than the relatively narrow c1 tooth (figs g and h).
In NZ there are are 2 forms of type IIA: the usual form, here called 'IIA narrow' and 'IIA broad'Ventromental plate ratio (VMR) - ratio of the width of the marginal region of ventromentum (usually seen as a granular band under light microscopy) (a in figure below) to the distance from the anterior margin to the base of the striae (b in figure below). 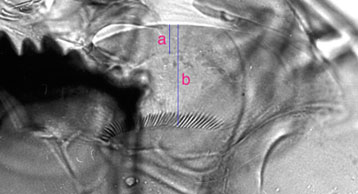 Pecten epipharyngis -
Proulx et al. (2013) recognised 4 types of PE in North American species. These are useful if the teeth are not worn down, as they often are in older larvae. Type D does not occur in any known Australian species.
Type A - fine sharp rather uniform teeth.
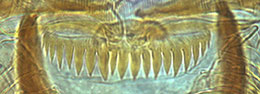
Type B - teeth broader but still sharp. Sometimes with one or two fine smaller teeth interspersed.
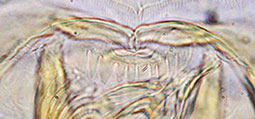
Type C - rounded and rather uniform. Worn type B teeth may be mistaken for this type.

Type D - rounded teeth with smaller teeth interspersed (generally found in the subgenera Lobochironomus or Chaetolabis).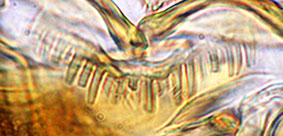 Premandible -
Assessment of the premandible can be difficult due to quite excessive wear and distortion in mounting. Where they are in good condition and lying flat, they can be categorised into 5 groups based on the relative widths of the teeth and whether they come to a sharp or a broad point. The teeth are measured as close as possible to their bases.
Premandible types
Type A: Both teeth narrow and coming to a fine point.
Type B: inner tooth moderately broad, about 3-5 times wider than, outer tooth. This can be split into 2 subgroups:
B1 Đ both teeth come to relatively fine points.
B2 Đ the inner tooth comes to a relatively broad point.
Type C: Both teeth are moderately broad, coming to relatively broad points.
Type D: Inner tooth very broad, outer tooth moderately broad.
Type E: Both teeth very broad and often quite short.
C. vitellinus has a six toothed premandible (C. javanus has five teeth).
Species of Kiefferulus also have a five-toothed premandible, but it is broader. Antennal ratio (AR) - measured on only the length of the sclerotized region of the segments, since the unsclerotized regions can expand to different extents in different specimens. Measured in this way the AR is usually higher. The mandible type is defined by the degree of darkening and separation of the 3rd inner tooth:
It seems better to consider the two characters separately.
Separation:
Type I - tooth fused
Type II - tooth partially free
Type III - tooth completely separated
Color:
Type A - tooth pale
Type B - some degree of pigmentation
Type C - as dark as other inner teeth
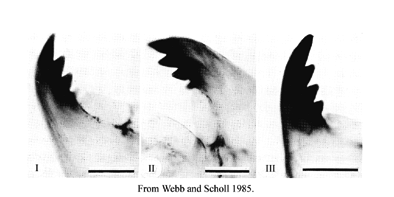
This figure shows IA; IIB; IIIC respectivelyMandible length and Mdt-Mat:
Hirvenoja and Michailova (1998) illustrated that the distance between the tip of the dorsal tooth and the apical tooth could differ between related species (Mdt-Mat) (blue line in figure below).
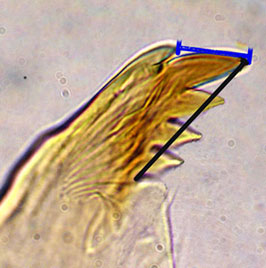
However for different sized species it may be preferable to divide this value by the length of the inner teeth (black line) to obtain the MTR.PACIFIC CHIRONOMUS
New Zealand has strong ties to the islands of the Pacific Ocean. This makes this listing a convenient place to record Chironomus species from these islands, although there appears to be no overlap of the Chironomid fauna (rather, many of them in fact are also found in South East Asia, so are also recorded in the listing of those species) Chironomus bicoloris Tokunaga 1964 Chironomus carolinense Tokunaga 1964 Chironomus circumdatus (Kieffer, 1916, as Tendipes) Chironomus claggi Tokunaga 1964 Chironomus crassicaudus Tokunaga 1964 Chironomus hawaiiensis Grimshaw 1901 Chironomus magnivalva Kieffer, 1917 Chironomus nigerilateralis Tokunaga 1964 Chironomus pallidinubeculosis Tokunaga 1964 Chironomus preapicalis Tokunaga 1964 Chironomus samoensis Edwards 1928 Chironomus sexipunctatus Tokunaga 1964 Chironomus trifasciatus Tokunaga 1964 Chironomus vitellinus Freeman 1961 Chironomus "harrisi" (sp. Fiji1)
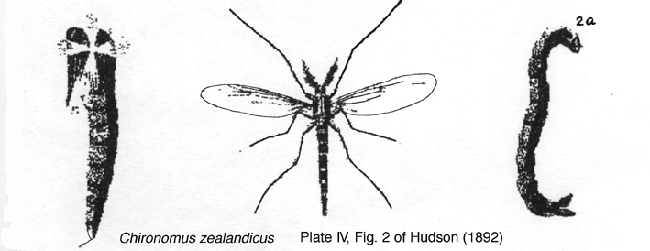
Chironomus zealandicus Hudson, 1892 There have been several attempts to relate C. zealandicus to one of the cytologically known species, and it has been suggested at different times that it was either species 1 (Forsyth, 1978 and Martin, 1998) or C. species a (Martin, 1996).
However, species 1 can be ruled out because there is an entry in Hudson's journal that he reared the specimens of C. zealandicus from larvae without ventral tubules. Forsyth therefore thinks that C. species a is the species that corresponds to Hudson's C. zealandicus. This is supported further by the fact that C. species a is the largest of the reared species, about the size of Hudson's C. zealandicus specimens.
The name is therefore used in this sense in this work.
Chromosome banding patterns have been published using the Australian standard system of Martin (1964), but are in process of being switched to the more universally used (although more difficult to uniquely specify bands) system of Keyl (1962) and Devai et al. (1989).
[ Go to References| RESEARCH ACTIVITES | Return to MARTIN LAB HOME ] |